|
Me and my partner testing what happens when you carbon dioxide to different temperatures of salt water (warm, cold, hot)
|
|
Title: The effect of water composition on the ph (absorption of CO2). Independent variable was the type of water. Dependent variable was the ph.
Topic Introduction: Ocean Acidification is the process of getting carbon dioxide from the atmosphere and putting it into the ocean that eventually is transformed into carbonic acid, which effects marine life.
Experimental Question:What are we trying to find out? How? Are trying to see what happens to seawater after adding carbon dioxide(CO2). Pre-Lab Question:1. What gas are you blowing into the water? The gas I am blowing into the water is carbon dioxide (CO2). 2. What happens to the gas when you blow it into the water? The gas, carbon dioxide will absorb the water making the water more acidic. 3. How are you measuring change in the water during this lab? I will be measuring the change in the water by color change 4. What does measuring the ph of the water tell us? The ph of the water tells us the acidity of the water. 5. After studying the reactions above, how do you think carbonic acid will affect the ph of salt water? Carbon dioxide(CO2) and water (H2O) makes carbonic acid (H2CO3).
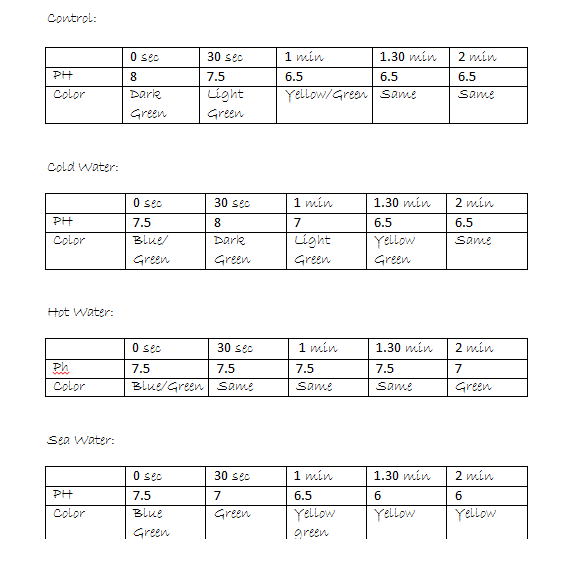
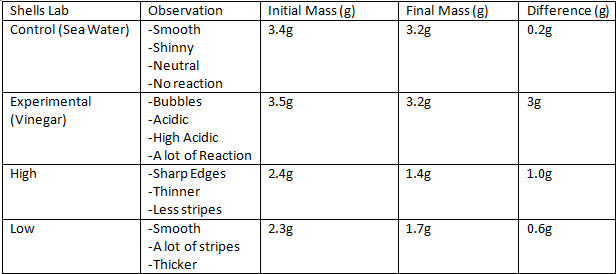
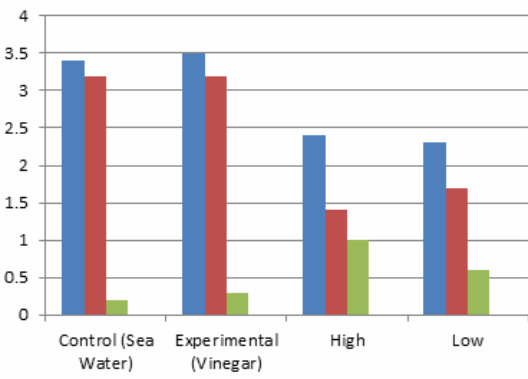
Hypothesis: I believe that the ph will get more acidic since we are adding carbon dioxide into the sea water. Hypothesis Shells Protocol : I believe that the shells will lose some color and have lost the hardness of the shell. Protocol: Control- The control of the experiment was the salt water. 100ml of the salt water was poured into a 200 ml beaker. Get 4 universal indicator, then wrap the top of the beaker with plastic wrap. Next was to poke a hole in the wrap to let the straw in, next my partner started to blow bubbles in the beaker for 2 minutes. Then repeat the steps. Our second choice was to use hot salt water, we did everything the same but the salt water was replaced with hot salt water.
Topic Introduction: Ocean Acidification is the process of getting carbon dioxide from the atmosphere and putting it into the ocean that eventually is transformed into carbonic acid, which effects marine life.
Experimental Question:What are we trying to find out? How? Are trying to see what happens to seawater after adding carbon dioxide(CO2). Pre-Lab Question:1. What gas are you blowing into the water? The gas I am blowing into the water is carbon dioxide (CO2). 2. What happens to the gas when you blow it into the water? The gas, carbon dioxide will absorb the water making the water more acidic. 3. How are you measuring change in the water during this lab? I will be measuring the change in the water by color change 4. What does measuring the ph of the water tell us? The ph of the water tells us the acidity of the water. 5. After studying the reactions above, how do you think carbonic acid will affect the ph of salt water? Carbon dioxide(CO2) and water (H2O) makes carbonic acid (H2CO3).
Hypothesis: I believe that the ph will get more acidic since we are adding carbon dioxide into the sea water. Hypothesis Shells Protocol : I believe that the shells will lose some color and have lost the hardness of the shell. Protocol: Control- The control of the experiment was the salt water. 100ml of the salt water was poured into a 200 ml beaker. Get 4 universal indicator, then wrap the top of the beaker with plastic wrap. Next was to poke a hole in the wrap to let the straw in, next my partner started to blow bubbles in the beaker for 2 minutes. Then repeat the steps. Our second choice was to use hot salt water, we did everything the same but the salt water was replaced with hot salt water.
Data Analysis : Post Lab Questions
1. As you blew through the straw, what were you adding to the water and how did that change the ph?
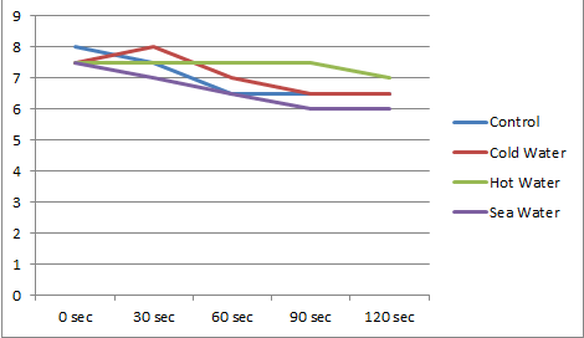
Carbon dioxide was being blown from the straw which made the ph more acidic. An depend on the temperature of the water determined how much Carbon Dioxide it can hold and how fast will it change.
2. What did the universal indicator tell us about the water?
The indicator told us the ph of the water.
3. What does this tell us about the effects of carbonic acid in ocean water?
When carbonic acid enters the oceans water it makes the water more acidic. An in places like the South/ North pole where the water is colder they will be able to hold more Carbon Dioxide when places like the Pacific Ocean where they already taken to much Carbon Dioxide which makes the water warmer it wont be able to hold that much.
4. Based on the results of your experimental protocol, which factor affects the ph of the water most, temperature or salt?
Based on the experimental protocol of the experimental protocol the factor that affects the ph of the water the most is temperature. Because colder water takes in more CO2 and warmer water takes in less CO2.
Conclusion:
Was your initial hypothesis correct? Why or why not? Explain. What did you learn about ocean acidification?
My initial hypothesis was correct. I said that the ph was going to get more acidic because we were adding carbon dioxide into the water and that's what happened. Any number from 1 to 7 is acid, the ph of the salt water was a constant rate of 7.5 but the more time passed it changed into 6.5. I learned that the carbon dioxide from the atmosphere traveling through into the ocean will have a dramatic change in ph levels which makes the oceans waters more acidic a harmful thing for the entire ecosystem.
1. As you blew through the straw, what were you adding to the water and how did that change the ph?
Carbon dioxide was being blown from the straw which made the ph more acidic. An depend on the temperature of the water determined how much Carbon Dioxide it can hold and how fast will it change.
2. What did the universal indicator tell us about the water?
The indicator told us the ph of the water.
3. What does this tell us about the effects of carbonic acid in ocean water?
When carbonic acid enters the oceans water it makes the water more acidic. An in places like the South/ North pole where the water is colder they will be able to hold more Carbon Dioxide when places like the Pacific Ocean where they already taken to much Carbon Dioxide which makes the water warmer it wont be able to hold that much.
4. Based on the results of your experimental protocol, which factor affects the ph of the water most, temperature or salt?
Based on the experimental protocol of the experimental protocol the factor that affects the ph of the water the most is temperature. Because colder water takes in more CO2 and warmer water takes in less CO2.
Conclusion:
Was your initial hypothesis correct? Why or why not? Explain. What did you learn about ocean acidification?
My initial hypothesis was correct. I said that the ph was going to get more acidic because we were adding carbon dioxide into the water and that's what happened. Any number from 1 to 7 is acid, the ph of the salt water was a constant rate of 7.5 but the more time passed it changed into 6.5. I learned that the carbon dioxide from the atmosphere traveling through into the ocean will have a dramatic change in ph levels which makes the oceans waters more acidic a harmful thing for the entire ecosystem.